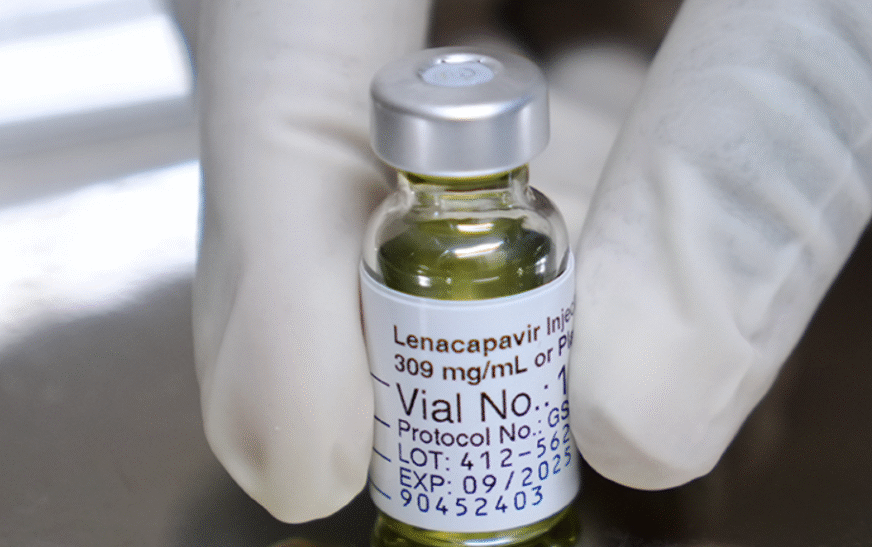Uganda Rolls Out Long-Acting HIV Prevention Injectables
Uganda has received its first consignment of 19,200 doses of Lenacapavir from the Global Fund, the Ministry of Health confirmed. “The drug is administered every six months to prevent HIV among people at substantial risk of infection. It will be distributed to high-burden and high-incidence districts starting March 2026,” the
Gov’t Approves Use of HIV injectables in Uganda
Uganda’s National Drug Authority (NDA) has approved the use of lenacapavir, a twice-yearly HIV prevention (PrEP) injection manufactured by Gilead Sciences, a U.S.-based company. The approval is a game-changer for HIV prevention, especially for people at high risk of infection. It marks a significant step toward ending AIDS by 2030.
New six months HIV Injectables cost Shs 100 million
The United States Food and Drug Administration (FDA) has approved long-acting injectable lenacapavir for HIV prevention. The new medicine is administered by injection once every six months and is a significant step in improving prevention options for people at risk of HIV infection around the world. In an interview, Gilead